How did the elemental composition of the universe evolve?
Why do we observe the types and quantities of chemical elements in the Universe that we do? This is one of the most fundamental questions facing astronomers. The field of nucleosynthesis studies how the elements were created, and it draws from a wide range of studies, from the Big Bang to the formation and evolution of our solar system.
Light elements (hydrogen, helium and lithium) were mainly created in the Big Bang. Measurement of the abundances of these elements can give scientists important clues to the nature and evolution of the early Universe. In particular, measurement of the ratio of the normal isotope of hydrogen to its heavier isotope known as deuterium provides important insight into primordial nucleosynthesis.
All other elements heavier than lithium are products of nuclear reactions occurring in stars and during supernova explosions. With certain exceptions, we have a comprehensive understanding of how a star evolves, as it converts hydrogen and helium into heavier elements. These heavy elements are disseminated into the Universe by stellar winds and supernova explosions. However, we need to understand just where and when the chemical evolution happens during the lifetime of a typical galaxy, and how well our models predict the observations.
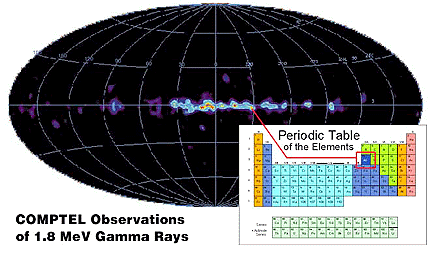
The study of radiation due to the decay of the products of nucleosynthesis provides direct information about the synthesis of elements. Due to its short lifetime (about 1 million years), 26 Aluminum is known to be an excellent tracer of the birthplace of elements. The 1.8 MeV 26Al emission line was shown to be concentrated on the galactic plane by observations of the COMPTEL instrument on NASA's Compton Gamma-Ray Observatory. Recent studies of this emission performed by GRIS have revealed spectral line broadening indicative of matter moving at about 500 km/second. This is much faster than any movement linked to the galactic rotation. Although 26Al can be released at high velocities to the interstellar medium by supernovae, it is hard to understand why it has not come to rest after a million years, the radiative lifetime. This observation constitutes a great challenge to our understanding of the origin and propagation of the 26Al.


