Solution for Student Worksheet: Energy Levels in the Atom
Solution for Calculate the Energy! Student Worksheet
The equation for determining the energy of any state (the nth) is as follows:
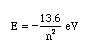
Answer the following questions:
1. Using the above expression, calculate the energy of the first excited state. Your answer will be negative. This signifies that the electron is bound to the atom (as opposed to being a free electron).
2 . Use the above expression to find the energy of the photon released when an electron around a hydrogen atom moves from the 4th to the 2nd level.
in the energies of the fourth (n=4) and second (n=2) levels
E = -13.6/42 - (-13.6/22)
E = -0.85 + 3.40
E = 2.55 eV
3. Now use the above expression to find the energy of the photon released when a free electron is captured to the 2nd level.
The energy of the photon emitted by a free electron captured to the n=2 level is thus
E = 0 - (-13.6/22) = 3.4 eV
4. Use the relationship between a photon's energy and its wavelength to calculate the wavelength of the photon emitted in question 2.
We can solve this for the wavelength, l = h c / E. where h = 6.626 x 10-34J-s, and c = 3 x 108 m/s.
We convert our energy E= 2.55 ev into Joules using 1 eV = 1.6x10-19 J. This gives an energy of E = 4.08 x 10-19 J.
We then find a wavelength of
l = ((6.626 x 10-34) x (3 x
108)) / (4.08 x 10-19)
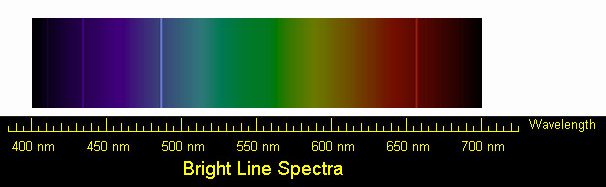
l = 4.87 x 10-7 m.
OR, using 1 nm = 1 x 10-9 m,
l = 487 nm.